Chlorine is found in the silicate mineral marialite.Ĭhlorine appears in the carbonate mineral northupite. Chlorine is a component with mercury in the sulfate mineral kleinite. It appears with copper in the sulfate mineral connellite. Chlorine appears with copper in atacamite.Ĭhlorine appears in the phosphate mineral chlorapatite. Chlorine is in the lead-containing carbonate mineral phosgenite. Other lead-containing chloride minerals are boleite, cumengite and laurionite. Lead with chlorine and fluorine form matlockite. A chloride of lead and antimony is nadorite, PbSbO 2Cl. A chloride of copper and lead is diaboleite. We assign half of this distance to each chlorine atom, giving chlorine a covalent atomic radius (r cov)Half the distance between the nuclei of two like atoms. From simple clear ionic crystals like halite, NaCl, to more complex halide containing minerals like the yellow crystals of mimetite, Pb 5(AsO 4) 3Cl, chlorides show a range of colors.One lead chloride mineral is mendipite, PbO 2Cl 2. This forms hydrochloric acid, a very strong acid.Ĭhlorine is a constituent of many mineral crystals. At 10C one volume of water dissolves 3.10 volumes of chlorine, at 30C only 1.77 volumes. The gas dissolves readily in water with the liberation of heat. It is a member of the halogen (salt-forming) group of elements and is obtained from chlorides by the action of oxidizing agents and more often by electrolysis it is a greenish-yellow gas, combining directly with nearly all elements. Hydrogen chloride, HCl, is a gas with a sharp unpleasant odor. This has led to widespread use in the sterilization of drinking water, for swimming pools, etc. It is manufactured on a large scale by electrolysis of concentrated solutions of sodium chloride.Ĭhlorine is a strong oxidizing agent and as a result is effective in killing bacteria. In pure form, it is a greenish-yellow gas with a sharp, irritating odor. It is not as chemically active as fluorine, but is still very active and forms compounds with most elements. O So far, you can rank the atomic radius of sulfur, chlorine, fluorine, and oxygen, in increasing order as: 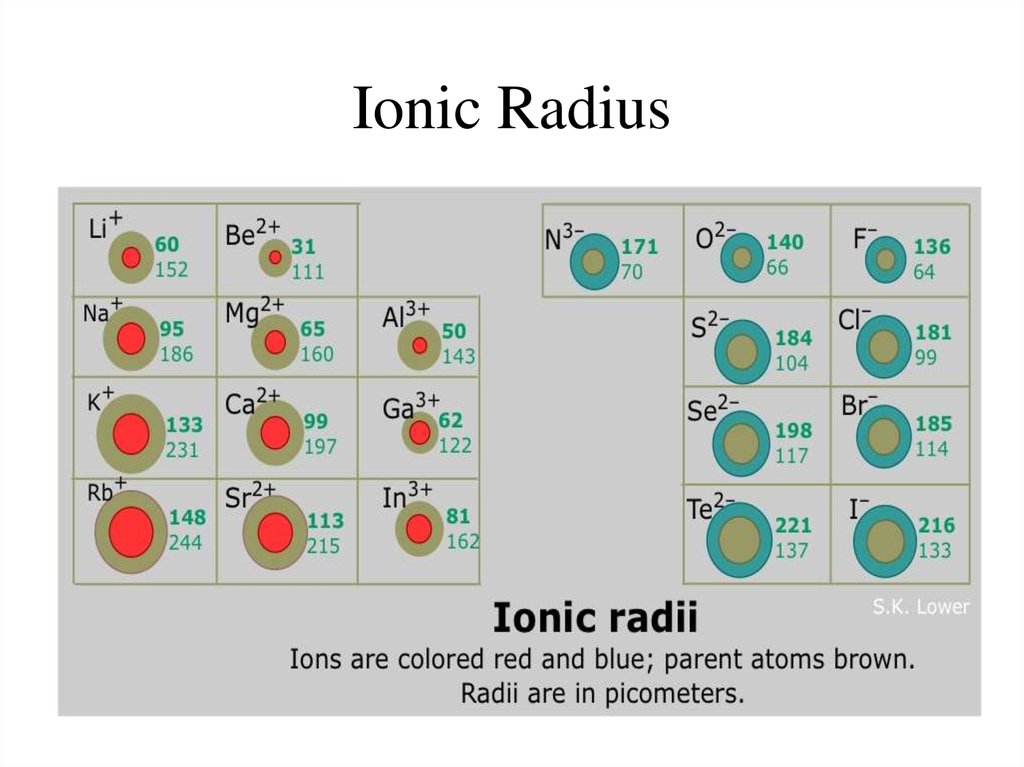
Sulfur and oxygen are in the same group (16), with sulfur directlly below oxygen, so sulfur the atomic radius of sulfur is larger than the atocmi radius of oxygen.Fluorine and chlorine are in the same group (17), with chlorine directly below fluorine, so the atomic radius of chlorine is larger than the atomic radius of fluorine.That permits you to compare the size of the elements in a group: Atomic radius increases from top to bottom within a group due to electron shielding. Sulfur and chlorine are in the period 4, being sulfur to the left of chlorine, so sulfur is larger than chlorine.Oxygen and fluorine are in the period 3, being oxygen to the left of fluorine, so oxygen is larger than fluorine. 
Hence, you can compare the elements that belong to a same period and predict that the atom with lower atomic number (number of protons) will haver larger atomic radius.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
